The Science of the Total Environment, 54 (1986)
207-216
CHEMICAL QUALITIES OF WATER THAT CONTRIBUTE TO HUMAN HEALTH
IN A POSITIVE WAY
HOWARD C. HOPPS
Medical College of Ohio, Department of Pathology, Toledo, OH
43699 (U.S.A.)
GERALD L. FEDER
U.S. Geological Survey, Lakewood, CO 80225
(U.S.A.)
ABSTRACT
The emphasis on harmful substances that may occur in potable
waters has almost obscured the fact that important beneficial
constituents are commonly present.
The chemical substances in water that make positive
contributions to human health act mainly in two ways: (i)
nutritionally, by supplying essential macro and micro elements
that the diet (excluding water) may not provide in adequate
amounts (for example, Mg, I and Zn); and (ii) by providing macro
and micro elements that inhibit the absorbtion and/or effects of
toxic elements such as Hg, Pb and Cd. Specific examples of these
beneficial effects will be given, also examples of harmful
effects on health that may result from excessive intake of these
ordinarily beneficial elements.
Because concentrations of the essential macro and micro
elements that occur in natural, potable waters vary greatly,
depending upon their source, geographic considerations are very
important in any studies attempting to relate water quality to
health. In this context, the inverse relationship between hard
water and cardiovascular disease will be discussed. Specific data
relating hardness and Mg and Ca content of potable waters to
specific geographic regions of the U.S.A. will be presented.
These data show a strong positive correlation between low Mg
content and decreased longevity, and between high Ca and Mg
content and increased longevity. In the regions considered,
increased longevity correlates strongly with decreased
cardiovascular mortality, and the decreased longevity with
increased cardiovascular mortality.
INTRODUCTION
Some toxic substances in water are not health threatening, at
least not to human beings. They may be toxic only to certain
species other than H. sapiens or, more important to our
considerations, toxic to H. sapiens, but ordinarily found at
concentrations insufficient to threaten human health. This
consideration influenced the title of our paper, with its
emphasis on contribution to health in a positive way,
implying that we shall not focus on the healthful aspects of
certain waters merely because they do not contain toxic
substances in health-threatening concentrations.
The importance of many natural, potable waters in human
nutrition has been largely ignored in the concern for
health-threatening, toxic substances that some waters contain. In
the context of positive contributions to human health, the
beneficial qualities of drinking water should also be
emphasized.
There are two major categories of these positive contributions
to health (aside from the obvious fact that water
(H2O) is essential to life and comprises about 60% of
the total body mass of the average normal adult human) —
nutritional and antitoxic.
Nutritional, through contributions of macro and micro
elements essential for health
These elements (Table 1), mainly ingested in the form of
inorganic ions, have many biological functions. For example, they
contribute to the formation of vitally important metalloenzymes,
carriers (e.g., hemoglobin), and selective membrane permeability;
and to the physical integrity of structures such as bone,
cartilage, and various fibrous materials (e.g., collagen and
elastica).

The practical significance of the first category depends upon
(a) the amount of the element in question that is often or
occasionally found in potable water, which varies greatly with
the geographic region; (b) whether or not the element, as it
occurs in potable water, is in a biologically active form (for
example, cobalt, as such, is not biologically active in terms of
human nutrition; to be nutritionally effective it must be
ingested and absorbed as a preformed complex, cobalamin); (c) the
extent to which the (regional) diet often or occasionally fails
to meet the individual's needs (this consideration must take into
account common physiologic and pathologic events that may require
amounts of the element that will exceed the recommended dietary
allowance (RDA) (NAS, 1980) for average humans).
Antitoxic contributions of certain macro and micro elements
that inhibit the adsorption and/or effects of toxic elements such
as mercury, lead and cadmium
These protective effects are, of course, limited, but may be
quite significant. Levander (1977) has reviewed much of the
evidence in support of this category. Stated very briefly,
protective action against toxicity from mercury is
provided by selenium; a high zinc intake appears to offer some
protection against the toxic effects of lead, whereas a
deficiency of calcium (also iron and copper to a lesser
extent) increases susceptibility to lead; selenium (also zinc and
iron to a lesser extent) decreases the toxicity of cadmium,
whereas a deficiency of calcium (also iron and copper to a lesser
extent) aggravates toxicity.
With respect to the nutritional qualities of potable waters,
we are mainly concerned with those macro and micro elements that
can be contributed in significant amounts by natural potable
waters, particularly if food (excluding water) may be inadequate
to provide the recommended daily allowances (RDA). The elements
that fall into this category are shown in Table 1.
Based upon an average consumption of 2 l of water per day for
adults (including the water content of coffee, tea, milk, fruit
juice, soft drinks, beer, soup, etc.) some natural potable waters
can supply all of the RDA for magnesium, fluorine, sodium,
iodine, and selenium, and more than one-third of the RDA of
calcium (Feder and Hopps, 1981). As much as 15% of the RDA of
zinc can also be supplied; this amount, although a relatively
small percentage of the RDA, can be quite significant because of
the wide spread deficiency of zinc in the United States. The
nutritional significance of chromium in drinking water has not
been demonstrated, but the predominant valance state of chromium
in natural potable waters (Cr3+ is the biologically
active state and, since the minimal requirement for absorbed
chromium is approximately 1µ per day (NAS, 1980), it is
reasonable to assume that some waters can provide nutritionally
significant amounts of this essential element. Chromium
deficiency is relatively common in the United States, and several
investigators have shown that it results in demonstrable states
of diabetes-like altered carbohydrate metabolism (Mertz, 1969;
Jeejeebhoy et al., 1977; Freund et al., 1979). Iron represents a
special case in that natural, nontoxic waters may supply more
than the RDA; however, waters containing concentrations of iron
in solution > 1 mg 1-1 are distasteful and are
rejected for aesthetic reasons.
Important consequences from deficiency of these elements may
become apparent only after many years and then, perhaps, only
when triggered by a pathologic event (for example,
deficiency of magnesium, when complicated by acute myocardial
ischemia, contributes to a lethal arrhythmia), or a physiological
stress (for example, deficiency of calcium. in association with
the post menopausal state and its associated endocrine
disturbances, contributes to osteoporosis). In experimental
animals, it is clear the deficiency of certain essential micro
elements early in life, or even during the fetal period, can lead
to a disease or disorder that manifests itself only many years
later — and perhaps this manifestation may be as subtle as
a relatively slight decrease in life span. Such delay is most
likely to occur when the deficiency is slight or moderate. These
observations imply that an adequate, balanced intake of essential
macro and microelements (coupled with otherwise good nutrition)
may increase life span compared with the average (Mertz, 1981). A
National Academy of Science sponsored study, Aging and the
Geochemical Environment (NAS, 1981), supports this view.
Although we are focusing upon ill effects that result from
deficiency of essential elements, it is important to point out
that too much of even an essential element can cause ill effects.
As in the case of deficiency, some times slight excesses may
require many years to manifest themselves in forms such as
increased risk of cancer, acceleration or exaggeration of
atherosclerosis, increased susceptibility to other "degenerative
diseases", and so on. The admonition of Paracelsus (1493-1541) is
very pertinent in this context: "All substances are poison; there
is none which is not a poison. The right dose differentiates a
poison and a remedy". Of the elements listed in Table 1, fluorine
and selenium deserve comment in this connection.
The effects of too much fluorine derived from natural drinking
waters are well known, ranging from slight cosmetic effects
(mottled tooth enamel) to incapacitating skeletal abnormalities.
Overt toxic effects from too much selenium also occur (rarely)
from ingestion of natural waters that contain high levels of
selenium, but, as with fluorine, occurrence is limited to small
geographic regions. Although selenium poisoning, i.e. selenosis
of livestock, has been recognized for many years (Underwood,
1977), human disease from naturally-occurring selenium poisoning
has been shown only recently; the occasional, poorly-documented
reports of past decades were not scientifically acceptable. Yang
et al., (1983) have reported selenium intoxication in the Hubei
Province of China involving several hundred individuals,
apparently the result of consuming vegetables with high
concentrations of Se in association with a low-protein diet.
Selenium intoxication in human beings from drinking water rich in
selenium has been reported in southwestern Colorado. On a Ute
reservation, some wells that provide drinking water for humans
contain very high concentrations of selenium — as high as
13000 µg 1-1 (Brogden et al., 1979). It is from
the consumption of very high selenium content waters that overt
manifestations of toxicity have been observed (Beath, 1962). It
may be that the toxicity of selenium to human beings has been
exaggerated because, in some areas of southwestern Colorado,
waters with 400-500 µg 1-1 of selenium have been
consumed by humans over many years without apparent harmful
effects (E.C. Hutchinson, U.S. Geological Survey, personal
communication, 1982). Data pertaining to selenium content of
drinking waters illustrate well the tremendous variation in
concentrations of trace elements in natural waters even within
relatively small geographic regions. In one 3-square mile area,
the Oxford Tract, selenium values in ground water ranged from
<10 to 13000 µg 1-1 (Brogden et al., 1979).
Vertical changes in Se concentrations are also important. In one
well that taps water at several depths on the Oxford Tract
installed by the U.S. Geological Survey, water samples collected
had selenium concentrations that ranged from 90 to 540 µg
1-1 (Brogden et al., 1979).
The most important benefit to human health associated with the
macro and micro elements contained in drinking water is probably
that related to cardiovascular mortality (CVM). Many
epidemiologic studies have shown an inverse relationship
between hard water and CVM, but the precise characteristics of
the "hard-water factor" remain elusive. Some (NAS, 1979) have
considered that substances contained in hard water may not be
directly responsible for decreased CVM; rather, soft
water may cause increased CVM because many soft waters
are corrosive and dissolve cadmium and/or lead from pipes that
distribute the water. In "Geochemistry of Water in Relation to
Cardiovascular Disease" (NAS, 1979), Comstock states". . . there
can be little doubt that the negative associations of water
hardness with cardiovascular mortality are not spurious", and he
evaluates several reports on the level of increased risk of
cardiovascular deaths in soft versus hard water areas in the
United States, Canada, and England and Wales. The data are not
entirely compatible because the grouping according to precise
cause of death varies, but the risk factor in the United States
appears to be about 1.15. This seems a weak association, but when
one considers that nearly 1 million persons in the U.S.A. die
each year from cardiovascular disease, approximately 2.5 times as
many as from cancer, an increased risk of death of 15% becomes
very significant indeed.
Many diseases contribute to deaths from cardiovascular disease
(CVM). Examples include certain developmental defects of the
heart, brain, or kidney; rheumatic fever; various other
infectious diseases; diabetes mellitus; hypertension; and, most
important of all, atherosclerosis. The decreased CVM associated
with water hardness appears to be most strongly related to
ischemic heart disease (NAS, 1981), the category which is
responsible for slightly more than half of all cardiovascular
deaths occurring in the United States. A weaker relationship
probably exists for stroke.
Virtually all diseases are multicausal, and ischemic heart
disease is no exception. Atherosclerosis of the coronary
arteries, which in itself has many causes, is the major
contributor to fatal ischemic heart disease. A thrombus imposed
upon an atherosclerotic plaque within a coronary artery, totally
blocking the flow of blood, is often the event that precipitates
sudden death; thus, blood-coagulation factors also play a role.
Furthermore, because the ultimate terminal event that causes
cessation of circulation is usually a cardiac arrhythmia
(including asystole), reactivity of the myocardium to electrical
stimuli is also an important factor. The predominant current view
is that hard water probably does not significantly affect the
degree or extent of atherosclerosis, but may influence
coagulative mechanisms and, more likely, may increase the
possibility that acute ischemic injury will result in fatal
cardiac arrhythmia (Aikawa, 1981).
The study we are reporting here is not primarily concerned
with the mechanisms by which hard water may reduce the risk of
death from certain cardiovascular diseases, but with specific
chemical substances in hard water that are associated with
decreased CVM and, perhaps, responsible for the decrease. It
addresses the question: how do hardness, calcium and magnesium
concentrations of potable ground waters correlate with CVM in two
large regions of the United States. The two contiguous regions
selected, each comprising more than 100 000 square miles, had
been identified and characterized in a previous National Academy
of Science sponsored study described in the monograph, "Aging and
the Geochemical Environment" (NAS, 1981). Longevity was
determined indirectly by measuring death rates (for natural
causes) of white males aged 35-74, age adjusted. The
difference in mortality of the two geographic regions was
striking. The decreased longevity region had a death rate nearly
twice as large as the region of increased longevity. The reader
is referred to the original source (NAS, 1981) for a detailed
description of the epidemiologic data (by H.I. Sauer), also the
reasons for the indirect method assessing longevity.
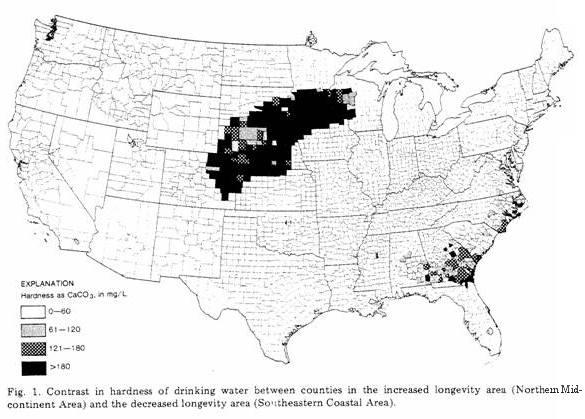
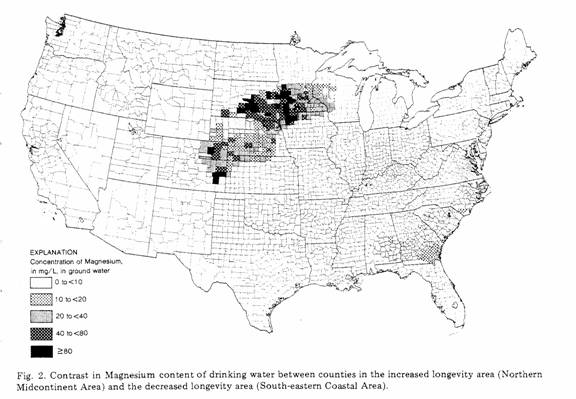
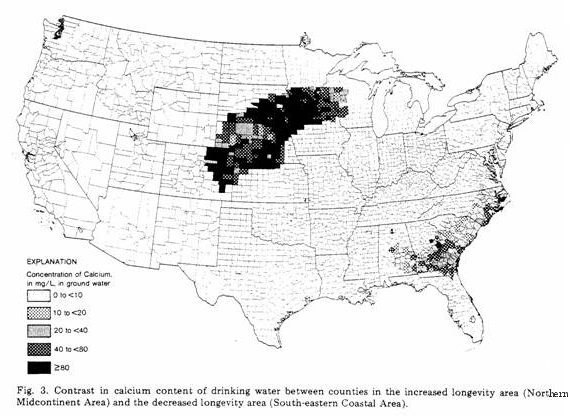
The two large areas identified in each of the maps shown as
Figs 1-3 represent the regions of increased longevity (Northern
Midcontinent Area) and decreased longevity (Southeastern Coastal
Area). The previous studies, referred to above, showed clearly
that the area of increased longevity was characterized by
markedly decreased CVM and, conversely, the decreased
longevity area by markedly increased CVM. In fact, CVM
was the major factor responsible for the increased and decreased
(compared with the average) longevity in the two regions. The
average water compositions for each county are shown for hardness
(Fig. 1) and for concentrations of magnesium (Fig. 2) and calcium
(Fig. 3). Magnesium and calcium were chosen for study, not only
because they are the most consistent, major cations present in
hard water, but also because one or the other or both are
generally regarded as the most probable cause of the hard water
effect on CVM (Marier et al., 1979). Hard water usually contains
considerable calcium, and may also contain considerable
magnesium. Limestone (CaCO3 and dolomite
(CaMg(CO3)2) are both major sources of
calcium in water. Of the two, only dolomite contributes
magnesium, however. Therefore, some natural hard waters have a
high Ca: Mg ratio, others a low Ca: Mg ratio, and many fall in an
intermediate range.
Figure 1 shows a striking, positive relationship between water
hardness and the region of increased longevity
(decreased CVM), and the converse relationship is also
quite good — that is, a negative relationship with the
region of decreased longevity (increased CVM). In Fig. 2
the relationship between low concentrations of magnesium
and decreased longevity (increased CVM) is very good, but the
converse is not as obvious. In Fig. 3 the high concentrations of
calcium correlate well with increased longevity (decreased CVM),
but the converse is questionable.
The decreased longevity (high CVM) region that we have studied
contains highly leached rocks and soil; this feature profoundly
influences the geochemical environment. The great majority of
potable waters there have very low total dissolved solids; all
cations and anions are in low concentrations. This characteristic
of many soft waters, coupled with the great variations of calcium
and magnesium concentrations (as well as other elements) among
hard waters makes it difficult to establish a correlation between
the concentration of a particular element and mortality from
cardiovascular disease, if indeed a prime component exists. At
any rate, it is unlikely that the hard-water factor will be
identified until more studies that relate cardiovascular
mortality to hardness of drinking water characterize chemically
the hard water and examine relationships between CVM and
specific chemical components of the hard water.
Given these caveats, our findings are compatible with the
hypothesis that magnesium at moderate concentrations provides a
modest degree of protection from CVM; thus, most hard waters will
satisfy this requirement, although other characteristics of the
water may overshadow magnesium, the prime component (?). Calcium
appears to provide additional protection from CVM. Thus,
geographic regions with very low CVM (compared with average CVM)
may best correlate with high-calcium content hard water, whereas
regions with very high CVM (compared with average rates) will
best correlate with very low Mg concentrations (e.g., very soft
waters).
SUMMARY
Many natural, potable waters contribute significant amounts of
macro and micro nutrients that are essential for human health.
Ordinarily, hard waters have greater nutritional benefits than
soft waters, but geographic variations are large, depending on
the geochemical environment. A major health benefit from drinking
hard water is a decreased risk of dying from cardiovascular
disease.
Two large contiguous geographic regions of the United States
— one characterized by higher than average cardiovascular
mortality (CVM), the other by lower than average CVM — are
compared in terms of the concentrations of hardness, and
magnesium and calcium in their natural, potable, ground waters.
Striking positive relationships exist between elevated hardness
and concentrations of calcium in water and decreased
CVM. In addition, the data show very good relationships between
softness and low concentrations of magnesium in water with
increased CVM. These findings are compatible with the
hypothesis that magnesium, in concentrations >20 mg
1-1, is the major factor in hard water responsible for
decreased risk of CVM. The data also suggest that calcium in
concentrations > 40-80 mg 1-1 provide additional
protection.
ACKNOWLEDGEMENT
This paper is an extension of material presented at the
"Medical Geology Conference on Health Threatening Toxins in
Water", 3 May 1984, directed by Dr E. Lynn Savage, Brooklyn
College, City University of New York.
REFERENCES
Aikawa, J.K., 1981. Magnesium: Its Biologic Significance. CRC
Press, Boca Raton, Florida.
Beath, O.A., 1962. Selenium poisons Indians. Sci. News Lett.
Si.: 254.
Brogden, R.E., E.C. Hutchinson and D.E. Hillier, 1979.
Availability and quality of ground water, Southern Ute
Reservation, Southwestern Colorado. U.S. Geological Survey
Water-Supply Paper 1576-J.
Feder, G.L. and H.C. Hopps, 1981. Variations in drinking water
quality and the possible effects on human health. In: D.D.
Hemphill (Ed.), Trace Substances in Environmental Health - XV.
University of Missouri, Columbia, Missouri, pp. 96-103.
Freund, H., S. Atamian and J.E. Fischer, 1979. Chromium
deficiency during total parenteral nutrition. J. Am. Med. Assoc.,
241: 496-498.
Jeejeebhoy, K.N., R.C. Chu, E.B. Marliss, G.R. Greenberg and
A. Bruce-Robertson, 1977. Chromium deficiency, glucose
intolerance, and neuropathy reversed by chromium supplementation,
in a patient receiving long-term total parenteral nutrition. Am.
J. Clin. Nutr., 30: 531-538.
Levander, O.A., 1977. Nutritional factors in relation to heavy
metal toxicants. Fed. Proc., Fed. Am. Soc. Exp. Biol., 36:
1683-1687.
Marier, J.R., L.C. Neri and T.W. Anderson, 1979. Water
Hardness Human Health, and the Importance of Magnesium. NRCC
Publication No. 7581, Ottawa, Ontario.
Mertz, W., 1969. Chromium occurrence and function in
biological systems. Physiol Rev., 49: 163-239.
Mertz, W., 1981. The essential trace elements. Science, 213:
1332-1338,
National Academy of Sciences (NAS), 1979. Geochemistry of
Water in Relation to Cardiovascular Disease. National Academy
Press, Washington, DC.
NAS, 1980. Recommended Dietary Allowances, 9th edn. National
Academy of Sciences, Washington, D.C.
NAS, 1981. Panel on Aging and the Geochemical Environment of
the U.S. National Committee for Geochemistry, The National
Research Council, Aging and the Geochemical Environment. National
Academy Press, Washington, D.C.
Underwood, E.J., 1977. Trace Elements in Human and Animal
Nutrition, 4th edn. Academic Press, New York.
Yang, G., S. Wang, R. Zhow and S. Sun, 1983. Intoxication of
humans in China. Am. J. Clin. Nutr., 37: 872-881.
This page was first uploaded to The Magnesium Web Site on
September 10, 2002
http://www.mgwater.com/




