American Journal of Epidemiology, Vol. 143, No. 5
Magnesium in Drinking Water and Death from Acute Myocardial
Infarction
Eva Rubenowitz, Gösta Axelsson, and Ragnar Rylander
Department of Environmental Medicine, Göteborg
University, Göteborg, Sweden
Reprint requests to Dr. Eva Rubenowitz, Department of
Environmental Medicine, Göteborg University, Medicinaregatan
16, 413 90 Göteborg, Sweden.
Abbreviations: CI, confidence interval: ICD,
International Classification of Diseases; pH, acidity as measured
by hydrogen ion concentration.
The relation between death from acute myocardial infarction
and the level of magnesium in drinking water was examined using
mortality registers and a case-control design. The study area
comprised 17 municipalities in the southern part of Sweden that
have different magnesium levels in the drinking water. Cases were
men in the area who had died of acute myocardial infarction
between ages 50 and 69 years during the period 1982-1989 (n =
854). The controls were men of the same age in the same area who
had died from cancer during the same time period (n = 989). In
both groups, only men who consumed water supplied from municipal
waterworks were included in the study. The subjects were divided
into quartiles according to the drinking water levels of
magnesium and calcium and the quotient between magnesium and
calcium. The odds ratios for death from acute myocardial
infarction in the groups were inversely related to the amount of
magnesium in drinking water. For the group with the highest
levels of magnesium in drinking water, the odds ratio adjusted
for age and calcium level was 0.65 (95 percent confidence
interval 0.50-0.84). There was no such relation for calcium. For
the magnesium/calcium quotient, the odds ratio was lower only for
the group with the highest quotient. These data suggest that
magnesium in drinking water is an important protective factor for
death from acute myocardial infarction among males. Am J
Epidemiol 1996;143:456-62.
Key words: calcium; coronary disease;
drinking water; magnesium; myocardial infarction
An inverse relation between water hardness and death from
cardiovascular disease has been observed in a number of studies
(1-7). A major hypothesis that has emerged from studies in recent
years is that magnesium, which together with calcium is the main
determinant of water hardness, protects against death from
ischemic heart disease. There are a number of facts to support
this hypothesis.
Magnesium is an enzyme activator, and is essential for
neuromuscular excitability and cell permeability. It is an
activator of sodium/potassium-adenosinetriphosphatase
(Na/K-ATP-ase), an enzyme that is necessary for the
transportation of sodium and potassium across cell membrane, and
that is thus necessary for maintaining the intracellular
concentrations of potassium (8, 9). It is also thought to
function as a natural calcium antagonist (9-12). Magnesium
deficiency can induce artery spasm, as has been shown in animal
experiments 13-16), and it facilitates the development of
arrhythmias (9, 12, 17). Significantly lower magnesium levels
have been found in the uninfarcted heart muscle of persons who
died suddenly from ischemic heart disease compared with persons
who died from other causes (18-23).
In the general population, the major portion of magnesium
intake is via food, and to a lesser extent via drinking water (in
Sweden, generally less than 5 percent). However, previous studies
support the hypothesis that magnesium in drinking water can be
critical for the magnesium content of the body. Relations have
been shown between water magnesium and magnesium content in heart
muscle (22), coronary arteries (24), and skeletal muscle
(25).
In the modern-day world, intake of dietary magnesium is often
lower than the recommended dietary amounts (6 mg/kg/day) (26).
For individuals at the borderline of magnesium deficiency,
waterborne magnesium can make an important contribution to the
total daily intake. In addition, the loss of magnesium from food
is lower when the food is cooked in magnesium-rich water. Another
reason why magnesium in water can play a critical role is its
higher bioavailability. Magnesium in water appears as hydrated
ions, which are more easily absorbed than magnesium in food
(27-29). The contribution of water magnesium among persons who
use water with high magnesium levels could thus be crucial in the
prevention of magnesium deficiency.
The absorption of magnesium is affected by simultaneous intake
of other nutrients. Examples of substances that can reduce the
absorption of magnesium are saturated fats, sugar, phytates,
proteins, phosphates, and calcium (26, 30). A quotient of 1/2
between magnesium and calcium has been suggested to be adequate
in the diet (26).
Epidemiologic studies in the United States (2), Canada (3 1,
32), South Africa (33), Finland (34,35), and Sweden (36) have
shown an inverse correlation between magnesium in drinking water
and mortality from ischemic heart disease. The relation has been
observed primarily among men.
It has been suggested that it is mainly the incidence of
sudden death from ischemic heart disease that is higher when
water magnesium levels are lower, owing, to an increased tendency
to vasoconstriction (15, 16) or arrhythmias (12, 32, 37, 38).
In the above-mentioned epidemiologic studies, except for those
done in Finland, the relation was studied on a group level and
the individual magnesium exposure was not determined. The aim of
this investigation was to study, on an individual level, the
relation between the amount of magnesium in drinking water and
death from myocardial infarction among males.
MATERIALS AND METHODS
A survey was carried out of the magnesium content in drinking
water in all Swedish municipalities. In part of southern Sweden,
it was found that, within a relatively small geographic area, the
water magnesium levels were different between
municipalities as well as within those municipalities. A
questionnaire was sent to all 37 municipal offices in two of the
counties in the area. Skåne and Blekinge (excluding the
city of Malmö), asking about drinking water hardness,
acidity (as measured by hydrogen ion concentration (pH)), and
water treatment procedures.
Of these 37 municipalities,17 were identified where the water
quality concerning the above parameters and treatment had been
basically unchanged (chance of hardness < 10 percent and pH
< 5 percent) during the last 10 years. Figure 1 shows the
location of the 17 municipalities that comprised the study
area.
Information on all men in the study area who had died between
ages 50 and 69 years during the period 1982-1989 was obtained
from the National Central Bureau of Statistics. Men with a
diagnosis of acute myocardial infarction (International
Classification of Diseases (ICD) code 410 (39)) as underlying
cause of death were considered cases. Controls were men with the
diagnosis of cancer (ICD codes 140-239) as the underlying cause
of death. The median age of death was 64 years in both
groups.
Parish population registers were used to obtain the addresses
of the study subjects. Information on the waterworks that
supplied the drinking water to each of the addresses, or the use
of private wells, was obtained from the municipalities.
According to the criteria described, 1,237 cases and 1,381
controls were identified.
The final numbers in the study were 854 cases and 989
controls. The numbers of persons excluded from the study and the
reasons for their exclusion are shown in table 1. The persons
excluded from the study were evenly distributed between cases and
controls, and over the study area.
Table 1.
Sample of myocardial infarction cases and controls, and the
losses according to exclusion criteria, in 17 municipalities,
southern Sweden, 1982-1989.
-----------------------------------------------------------------
No. of No. of
cases
controls*
-----------------------------------------------------------------
Initial sample 1,237 1,381
Insufficient information on
addresses or waterworks 129 117
Less than one year at the last address 36 64
Used own well 218 211
Final sample 854 989
----------------------------------------------------------------
* Controls were men of the same age (50-59 years) in the same
study area as the cases and who had died of cancer.
Information on levels of magnesium and calcium in tap water
was obtained from each waterworks. Table 2 shows the number of
waterworks, the range of magnesium in drinking water, and the
amount of magnesium in water supplied to the most densely
populated area in each municipality. Within the municipalities,
the number of waterworks engaged in the study varied between 1
and 15, and the total number was 78. The amount of magnesium in
the drinking water in the municipalities varied between 1.3 and
20.0 mg/liter, and the amount of calcium ranged from 22 to 225
mg/liter. There was no correlation between magnesium and calcium
in the drinking water (rxy = 0.056). Water magnesium in
the most densely populated area varied between 1.3 and 20.0
mg/liter.
TABLE 2.
Number of waterworks, the range of magnesium in drinking water,
and the amount of magnesium in water supplied to the most
densely populated areas of 17 municipalities, southern Sweden,
1982-1989.
-----------------------------------------------------------------
Range of Water Magnesium
Municipality No. of magnesium (mg/liter) in the
no.* waterworks in water most densely
in the study (mg/liter) populated area
-----------------------------------------------------------------
1 2 5.9-10.0 10.0
2 2 6.8-20.0 20.0
3 4 2.6-10.0 9.0
4 2 8.8-13.0 8.8
5 5 5.1-11.9 7.5
6 4 6.5-18.0 16.0
7 9 3.3-13.5 5.0
8 3 3.0-6.9 6.9
9 5 1.3-7.6 1.3
10 2 4.0-9.0 9.0
11 2 5.5-9.0 8.0
12 1 10.0 10.0
13 15 1.3-14.4 6.7
14 9 3.8-13.4 9.7
15 2 7.0-11.0 11.0
16 9 2.0-13.0 2.0
17 2 1.9-5.0 2.0
----------------------------------------------------------------
* Municipality numbers correspond to figure 1.
A measure of validity of the data for the water magnesium
content was made. One sample of tap water, taken at the
municipality office, was obtained from the main waterworks of
each municipality. The sample was analyzed with atomic absorption
spectrophotometry at the Chalmer's University of Technology,
Göteborg, Sweden. There was a strong correlation between the
values of magnesium and calcium in these analyses and the values
we obtained from the waterworks (magnesium: rxy = 0.96,
p < 0.001; calcium: rxy = 0.97, p < 0.001). This
demonstrated that the content of magnesium in the water did not
change on the way from the waterworks to the households.
Logistic regression models were fitted to the data with the
EGRET software package (SERC, Seattle, Washington) for
unconditional maximum likelihood estimation of the regression
parameters. In the analyses, the subjects were divided into
quartiles according to the levels of magnesium, calcium, and
magnesium calcium ratio in the drinking water. Odds ratios were
calculated in relation to the group with the lowest exposure. In
addition, trend analyses with 2 X K tables were made. Adjustments
for age were made in all analyses. Values of p < 0.05 were
considered statistically significant.
RESULTS
Table 3 shows the numbers of cases and controls and odds
ratios in relation to the magnesium content in drinking
water.

The odds ratios for death from acute myocardial infarction
were significantly lower for the two groups with high levels of
magnesium in the drinking water. Adjustments for the amount of
calcium in drinking water only slightly altered the odds ratios.
The adjusted odds, ratio (95 percent confidence interval (Cl))
was 0.88 (0.66-1.16) for the group with water magnesium levels
between 3.6 and 6.8 mg/liter, 0.70 (0.53-0.93) for the group with
magnesium levels between 6.9 and 9.7 mg/liter, and 0.65
(0.50-0.84) for the group with magnesium levels of 9.8 mg/liter
or more. Trend analyses showed statistical significance for
magnesium adjusted for calcium.
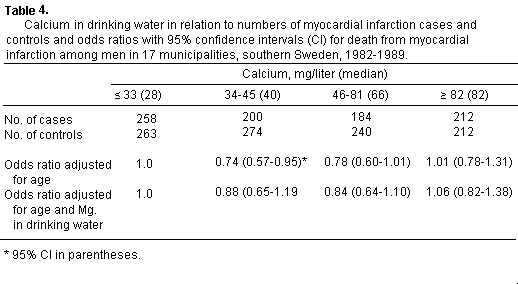
Odds ratios in relation to calcium levels showed slightly
lower values with higher calcium levels, but when adjusted for
magnesium there was no difference between the groups with
different levels of calcium (table 4). Trend analyses showed no
statistical significance for calcium.
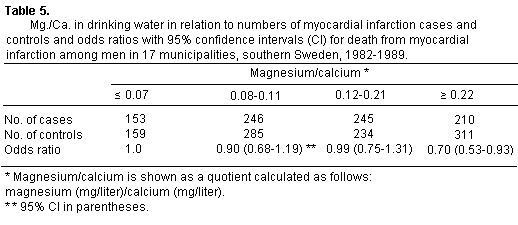
The odds ratios in relation to magnesium/calcium quotient are
shown in table 5. The group with the highest quotient (= or >
0.22) had a significantly lower odds ratio (0.70, 95 percent Cl
0.53-0.93). Odds ratios for the other groups were not
significantly lower. The quotient for magnesium/calcium in water
showed a trend similar to that seen for magnesium alone (p <
0.05). Adjustments for age, which were made in all analyses, only
affected the odds ratios slightly.
DISCUSSION
Epidemiologic studies on the group level involve several
methodological difficulties, particularly the description of the
dose. In contrast to several previous investigations, this study
determined the source of drinking water on the individual level,
taking into account the fact that many municipalities were using
several different waterworks, and avoiding the possible
interference of private wells.
Most of the waterworks had information on magnesium levels
only for the most recent years (1990-1991). Because only those
municipalities were chosen where the water source, quality, and
treatment had been stable for the last 10 years, we made the
assumption that the magnesium levels in 1990-1991 were about the
same as during the period when the mortality data were collected
(1982-1989). The assumption is supported by the fact that calcium
levels which had been registered for a longer time, had been
stable over the time period. In the analyses, we used the values
of calcium observed during the same period as that of the
magnesium values.
Although the study offered an increase in precision in regard
to the exposure to magnesium on the individual level, we were
unable to calculate the exact amount of magnesium taken in from
water, because the amounts of water consumed at home or at other
places and the use of water filters could not be determined. The
data on individual exposure were thus still characterized by a
lack of precision.
As with all retrospective register studies, this study suffers
from some uncertainty regarding the accuracy of diagnosis. In the
study, death certificates formed the basis for the diagnosis.
Autopsies were carried out in 40.6 percent of cases and in 23.6
percent of controls, and hospital examination before death in
50.1 percent and 75.4 percent, and medical examination outside
hospital before death or examination after death in 9.3 percent
and 1.0 percent, respectively. This allowed some validation of
diagnosis. The autopsy frequency was nearly the same over the
study area and in the four quartiles of magnesium in water
(cases, 43, 37, 41, and 42 percent, respectively; controls, 25,
25, 24, and 21 percent, respectively).
We only included those persons who had lived at their address
for at least a year at the time of death. Some investigators have
found an influence of magnesium on the extent of atherosclerosis
(37) and, if that is so, the exposure of water magnesium further
back in time would be of interest. However, Crawford and Crawford
(24) could not detect any difference in the prevalence of
atherosclerosis between residents of hard-water and soft-water
areas, although the levels of magnesium were lower in coronary
arteries among persons in soft-water areas. The possible role of
magnesium in preventing atherosclerosis is probably of minor
importance in comparison to the predominant effect -- the
prevention of sudden death from ischemic heart disease, owing to
its influence on blood vessel contraction and arrhythmia, which
is related to the current magnesium status. A one-year exposure
period would thus be relevant for this type of study because it
is well sufficient for an adaptation to the local water magnesium
level (26).
The controls were men who had died from cancer. To our
knowledge, there are no data which suggest that magnesium could
influence the risk of death from cancer. However, for several
other common diseases, such as cerebrovascular and respiratory
disease, there are data showing a possible relation with
magnesium (2, 40). Accidental death is not a suitable reference
diagnosis because in such cases of death alcohol is often
involved, and it is known that alcohol affects the magnesium
status (41).
Chlorinated surface water has been associated with cancers of
the bladder and pancreas (42, 43). Among the 989 controls in the
study, bladder and pancreatic cancers were the underlying cause
of death among 28 and 66 controls, respectively.
Only three waterworks of 78 in the study supplied chlorinated
surface water. Nine waterworks supplied chlorinated ground water.
The magnesium levels in the water from the waterworks with
chlorinated water ranged between 2.2 and 13.0 mg/liter, thus
representing all quartiles. There is thus no reason to believe
that this would be a confounding factor.
The strong negative relation found in this study, between
death from myocardial infarction and drinking water magnesium,
supports the results of several previous studies (2, 31-36). A
few studies have reported contradictory results. Shaper et al.
(44) found no relation between magnesium and cardiovascular
mortality in Great Britain, although a significant relation was
found for water hardness. However, their study included all
cardiovascular diseases and did not distinguish between ischemic
heart disease and stroke. As discussed above, it is death from
ischemic heart disease that is of interest in regard to
magnesium, and, furthermore, an inverse relation between calcium
and cerebrovascular disease has been shown in previous studies
(36).
Nerbrand et al. (7) studied water quality in 76 municipalities
in Sweden in relation to cardiovascular mortality in 1969-1983.
Water hardness was measured in 1980 and correlated negatively to
ischemic heart disease mortality. Magnesium was measured 10 years
later, in 1990, and no relation to magnesium was found. However,
only average values of water magnesium in the municipalities were
used, instead of considering the value from each waterworks, and
those municipalities where water quality and treatment had been
changed since 1980 were not excluded.
Biörk et al. (45) found a relation between water hardness
and mortality from "arteriosclerotic heart disease" and "other
degenerative heart diseases" but no relation to magnesium. In
that study, however, magnesium data were available only from 14
of the 34 cities from which the populations were selected.
Moreover, the largest cities in Sweden were included, and
cardiovascular deaths in large cities involve a complex
interaction in which life-style factors play a large role.
Thus, in our view, these studies with contradictory findings
have important shortcomings in study design that make the
conclusions less reliable.
In this study, the municipalities that were included were all
middle-sized, and the area is geographically and culturally
homogeneous. There is thus no reason to believe that there would
be any correlation between such risk factors as smoking, fat
consumption, and stress and the level of magnesium in drinking
water in the study area, especially since the levels of magnesium
varied within the municipalities as well as between them (table
2). Besides, the magnesium levels in the most densely populated
area in the municipalities represented the whole range of levels
(table 2).
The results show that magnesium alone was more important for
the risk of having an acute myocardial infarction than was the
magnesium/calcium quotient. The quotient of 1/2 discussed above
in regard to food seemed to be less important in drinking
water.
The results from this study strengthen the hypothesis that
magnesium in drinking water helps to prevent death from
myocardial infarction. An attempt to quantify the importance of
magnesium has been made by Marier and Neri (46), using a number
of the above-mentioned epidemiologic studies. They estimated that
an increase in water magnesium level of 6 mg/liter would decrease
ischemic heart disease mortality by approximately 10 percent. The
data from this study could be used to estimate the impact of
magnesium on the incidence of myocardial infarction in the study
population. If everyone were to drink water from the highest
quartile (= or > 9.8 mg/liter), the decrease in mortality from
acute myocardial infarction would be about 19 percent . This
means that the age-specific incidence of death from myocardial
infarction in the study area would change from about 350/100.000
to 285/100.000. The incidence decrease per mg/liter magnesium
could be calculated to be approximately 10/100.000, which is an
even larger decrease than Marier and Neri (46) estimated.
Future studies should increase the precision of the estimation
of the individual's intake of magnesium both via food and water,
and control for confounding factors. This could be done in
prospective studies on cases of non-fatal myocardial infarction.
In such studies, it would also be possible to investigate whether
magnesium in drinking water is important for people in general,
or if there are certain risk groups that benefit from high
magnesium levels in drinking water.
ACKNOWLEDGMENTS
The study was supported by funds from the Swedish Environment
Protection Agency (grant no. 38405), the Göteborg Medical
Society, and the research funds of Clas Groschinsky, Salus, and
Konrad and Helfrid Johansson.
The authors acknowledge with gratitude the help from the staff
at the parish population registers and the municipalities for
tracing the addresses and waterworks. They also wish to thank
Prof. Lars Wilhelmsen for valuable advice.
REFERENCES
1. Kobayashi J. On geographical relations between the chemical
nature of river water and death rate from apoplexy. Berichte des
Ohara Instituts für Landwirtschaftliche Biologie, Okayama
University 1957;11:12-21.
2 . Schroeder HA. Relationship between mortality from
cardiovascular disease and treated water supplies. Variations in
states and 163 largest municipalities of the United States JAMA
1960;172:1902-8.
3. Crawford MD, Gardner MJ, Morris, JN. Mortality and hardness
of local water supplies. Lancet 1969;1:827-31.
4. Comstock GW. Water hardness and cardiovascular disease. Am
J Epidemiol 1979;110:375-400.
5. Anderson TW, Le Rich LH, MacKay JS. Sudden death and
ischemic heart disease: correlation with water hardness and local
water supply. N Engl J Med 1969;280:805-6.
6. Perry HM, Perry EF. Cardiovascular disease: the effect of
hard and soft water. In: Boström H, Ljungstedt N, eds.
Skandia International Symposia: Trace Elements in Health and
Disease. Stockholm: Almqvist & Wiksell International,
1985;13:193-220.
7. Nerbrand C, Svärdsudd K, Ek J, et al. Cardiovascular
mortality and morbidity in seven counties in Sweden in relation
to water hardness and geological settings. Eur Heart J
1992:13:721-7.
8. Günther T. Magnesium: cardiovascular biochemistry.
Magnesium Bull 1986;8:136-9.
9. Reinhardt RA. Clinical correlates of the molecular and
cellular actions of magnesium on the cardiovascular system. Am
Heart J 1991;121:1513-21.
10. Levine BS, Coburn JW. Magnesium -- the mimic antagonist of
calcium. N Engl J Med 1984;310:1253-5.
11. Sjögren A, Edvinsson AL. The influence of magnesium
on the release of calcium intracellular depots in vascular smooth
muscle cells. Pharmacol Toxicol 1988;62:17-21.
12. Sjögren A, Edvinsson L. Fallgren B. Magnesium
deficiency in coronary artery disease and cardiac arrhythmias. J
Int Med 1989;226:213-22.
13. Altura BM, Altura BT, Gebrewold A, et al. Magnesium
deficiency and hypertension-correlation between magnesium
deficient diets and microcirculatory changes in situ. Science
1984;223:1315-17.
14. Luthringer C, Rayssiguier Y. Guera E, et al. Effect of
moderate magnesium deficiency on serum lipids, blood pressure and
cardiovascular reactivity in normotensive rats. Br J Nutrition
1988;59:243-50.
15. Turlapaty PDMV, Altura BM. Magnesium deficiency produced
spasms of coronary arteries: relationship to etiology of sudden
death ischemic heart disease. Science 1980;208:198 -200.
16. Altura BM, Altura BT, Carella A. et al. Hypomagnesemia and
vasoconstriction: Possible relationship to etiology of sudden
death ischemic heart disease and hypertensive vascular disease.
Artery 1981;9:212-31.
17. Eisenberg MJ. Magnesium deficiency and cardiac
arrhythmias. N Y State J Med 1986;86:133-5.
18. Heggtveit HA, Tanser P, Hunt B. Magnesium content of
normal and ischemic human hearts. 7th International Congress of
Clinical Pathology, Montreal, Québec, July 13-19,
1969:53.
19. Chipperfield B, Chipperfield JR. Heart muscle magnesium,
potassium and zinc concentrations after sudden death from heart
disease. Lancet 1973;2:293-6.
20. Chipperfield B, Chipperfield JR. Differences in metal
content of the heart muscle in death from ischemic heart disease.
Am Heart J 1978;95:732-7.
21. Behr G, Burton P. Heart-muscle magnesium. (Letter). Lancet
1973;2:450.
22. Anderson TW, Neri LC. Schreiber GB, et al. lschemic heart
disease, water hardness and myocardial magnesium. Can Med Assoc J
1975;113:199-203.
23. Elwood PC. Myocardial magnesium and ischaemic heart
disease. Artery 1981;9:200-4.
24. Crawford T, Crawford MD. Prevalence and pathological
changes of ischaemic heart disease in hard-water and in a
soft-water area. Lancet 1967;2:229-32.
25. Landin K, Bonevik H, Rylander R, et al. Skeletal muscle
magnesium and drinking water magnesium level. Magnesium Bull
1989;11:177-9.
26. Durlach J. Recommended dietary amounts of magnesium: Mg
RDA. Magnesium Res 1989;2,3:195-203.
27. Durlach J, Bara M, Guiet-Bara A. Magnesium level in
drinking water: its importance in cardiovascular risk. In:
Itokawa Y, Durlach J, eds. Magnesium in health and disease.
London: John Libbey & Co Ltd. 1989:173-82.
28. Theophanides T, Angiboust J-F, Polissiou M, et al.
Possible role of water structure in biological magnesium systems.
Magnesium Res 1990;3:5-13.
29. Löwik MRH, Groot EH. Binnerts WT. Magnesium and
public health: the impact of drinking water. In: Trace substances
in environmental health: proceedings of University of Missouri's
annual conference on the trace substances in environmental
health. 1982;16:189-95.
30. Seelig MS. Nutritional status and requirements of
Magnesium: with consideration of individual differences and
prevention of cardiovascular disease. Magnesium Bull
1986;8:170-185.
31. Anderson TW, Le Riche WH. Sudden death from ischemic heart
disease in Ontario and its correlation with water hardness and
other factors. Can Med Assoc J 1971;105:155-60.
32. Allen HAJ. An investigation of water hardness, calcium and
magnesium in relation to mortality in Ontario. PhD thesis.
Ontario: University of Waterloo, 1972.
33. Leary WP, Reyes AJ, Lockett CJ, et al. Magnesium and
deaths ascribed to ischaemic heart disease in South Africa. S Afr
Med J 1983;64:775-6.
34. Punsar S, Karvonen MJ. Drinking water quality and sudden
death: observations from West and East Finland. Cardiology
1979;64:24-34.
35. Luoma H, Aromaa A, Helminen S, et al. Risk of myocardial
infarction in Finnish men in relation to fluoride, magnesium and
calcium concentration in drinking water. Acta Med Scand
1983;213:171-6.
36. Rylander R, Bonevik H, Rubenowitz E. Magnesium and calcium
in drinking water and cardiovascular mortality. Scand J Work
Environ Health 1991;17:91-4.
37. Seelig MS, Heggtveit HA. Magnesium interrelationships in
ischemic heart disease: a review. Am J Clin Nutr 1974;27;
59-79.
38. Anderson TW. Water hardness, magnesium and ischemic heart
disease. Nova Scotia Med Bull 1977;56:58-61.
39. World Health Organization. International classification of
diseases. 9th revision. Geneva: World Health Organization,
1977.
40. Mathew R, Altura BM. Magnesium and the lungs. Magnesium
1988;7:173-87.
41. Flink EB, Omar M, Shane SR. Alcoholism, liver disease and
Mg. Magnesium Bull 1981;3:209-16.
42. Neutra RR, Ostro B. An evaluation of the role of
epidemiology in assessing current and future disinfection
technologies for drinking water. Sci Total Environ 1992;
127(1-2):91-122.
43. IJsselmuiden CB, Gaydos C, Feighner B, et al. Cancer of
the pancreas and drinking water: a population-based case-control
study in Washington County, Maryland. Am J Epidemiol
1992;136:836-42.
44. Shaper AG, Packham RF, Pocock SJ. The British Regional
Heart Study: cardiovascular mortality and water quality, J
Environ Pathol Toxicol 1980;4-2,3:89-111.
45. Biörck G, Boström H, Widström A. On the
relationship between water hardness and death rates in
cardiovascular diseases. Acta Med Scand 1965;178:239-52.
46. Marier JR, Neri LL. Quantifying the role of magnesium in
the interrelationship between human mortality/morbidity and water
hardness. Magnesium 1985;4:53-59.
This page was first uploaded to The Magnesium Web Site on June
12, 1996
http://www.mgwater.com/




