THE MAGNESIUM REPORT
Clinical, Research, and Laboratory News for Cardiologists
First Quarter 1999
Oral Magnesium and Hypertension: Research and Clinical
Application
LAWRENCE M. RESNICK, MD
Knowledge of the association between magnesium and
hypertension is almost a century old, beginning with the
observations that parenteral magnesium sulfate reduced blood
pressure (BP) in eclampsia and pre-eclampsia, and also in
non-pregnant subjects with malignant hypertension. More recently,
further observations support the BP-magnesium linkage:
Epidemiologically, multiple studies have identified an inverse
relationship between dietary magnesium intake and BP.
Pathophysiologically, cellular depletion of magnesium contributes
to, and is a feature shared in common by subjects with
hypertension, left ventricular hypertrophy (LVH), insulin
resistance, obesity, and frank diabetes mellitus. This
constellation of vascular and metabolic conditions is known
variously as “syndrome X,” generalized
cardiovascular-metabolic disease, or the plurimetabolic syndrome.
Therapeutically, both animal studies and clinical trials have
demonstrated that oral magnesium may exert a beneficial effect on
hypertension.
Altogether, data now suggest that magnesium may be ready for
“prime time” in hypertension management. The
question-and-answer feature (below) describes in greater detail
how oral magnesium may be used to treat appropriate patients with
hypertension.
Epidemiologic studies
Ascherio and colleagues at Harvard examined the dietary habits
of approximately 40000 nurses and 30000 male health professionals
and found lower BPs in subjects consuming more magnesium (as
fruits and vegetables). Similarly, of various dietary factors
analyzed in the Honolulu Heart Study, magnesium was the strongest
correlate of both systolic and diastolic BP among 615 men of
Japanese ancestry living in Hawaii. Furthermore, in 14000
middle-aged adults followed for up to 7 years in the
Atherosclerosis Risk in Communities (ARIC) Study, lower serum
magnesium levels were associated with an increased risk of
coronary heart disease and future hypertension and also
quantitatively predicted the rate of atherosclerotic plaque
accumulation in the carotid artery—the lower the magnesium
level, the worse the outcome.
Magnesium-calcium link
Over 40 years ago, Altura and coworkers demonstrated that
lower magnesium levels in vitro cause vasoconstriction, while
increased magnesium dilates blood vessels and blocks the
vasoconstrictor effects of substances such as epinephrine. In
vivo, dietary magnesium deficiency itself may raise BP.
About 15 years ago, using nuclear magnetic spectroscopy, our
group observed an intracellular magnesium deficit in human
hypertension. Working with biophysicist Raj K. Gupta, we noted
that BP among all subjects, whether hypertensive or not, is
inversely related to basal fasting intracellular free magnesium
levels—the lower the free magnesium level, the higher the
BP. This demonstration parallels a Swiss report published the
same year, showing the opposite relation for calcium—the
higher the intracellular free calcium, the higher the BP. These
clinical observations are relevant since in vitro studies have
long observed opposite tissue effects of each mineral ion on the
other—loading tissues with calcium depletes magnesium, and
vice versa.
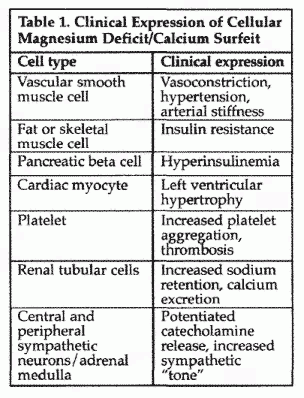
We subsequently showed this constellation of suppressed
intracellular free magnesium and/or elevated intracellular free
calcium levels not only in hypertension, but also in other
conditions associated with it, such as arterial stiffness
(decreased compliance), LVH, insulin resistance,
hyperinsulinemia, and frank diabetes mellitus—all
components of what has become known as the metabolic syndrome X.
This led us to propose the “ionic hypothesis of
cardiovascular and metabolic disease” in which each of the
conditions mentioned above is linked by and represents different
tissue-specific expressions of the same underlying cellular
lesion, characterized at least in part by suppressed
intracellular free magnesium and elevated cytosolic free calcium
levels (Table 1).
Rationale for using oral magnesium
Based on the above epidemiologic, dietary, experimental, and
clinical research, it seems reasonable to consider the use of
oral magnesium supplementation by either dietary or pill
supplementation as a therapeutic option in subjects with these
common cardiovascular and metabolic conditions. This is
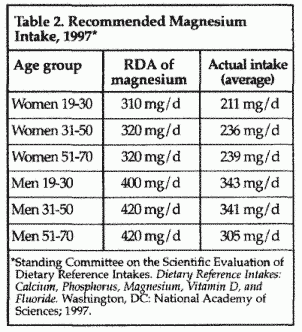
especially relevant since currently, both men and women consume
considerably less than the recommended dietary allowance (RDA) of
magnesium (Table 2). Indeed, over the course of this century the
per capita dietary intake of magnesium in the United States has
decreased by 50%.
The cumulative experience from published clinical trials of
magnesium supplementation in hypertension has been encouraging.
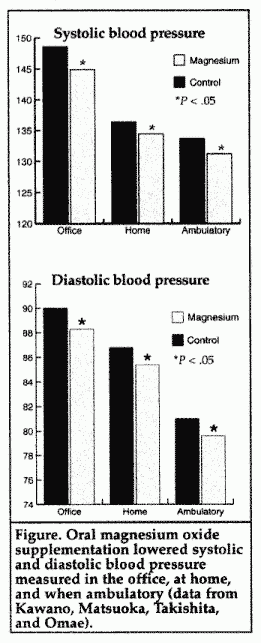
Kawano and colleagues recently studied the effects of 8 weeks of
magnesium oxide, 400 mg twice daily (20 mmol or 480 mg of
elemental magnesium), on office, home, and ambulatory BP in 34
male and 26 female Japanese subjects with essential hypertension,
ages 33-74. Both diastolic and systolic BPs fell significantly
during supplementation; those having the highest baseline BPs
experienced the greatest magnesium-induced fall in pressure
(Figure). Similarly, Itoh and colleagues found that oral
magnesium supplementation for 4 weeks (548 mg/d as magnesium
hydroxide to male subjects and 411 mg/d to female subjects) not
only increased serum levels of magnesium, but lowered BP and
improved total cholesterol and high to low density lipoprotein
(HDL:LDL) ratios in otherwise healthy
volunteers.
Supporting these reports are various others suggesting the
utility of magnesium supplementation in hypertension. Witteman
and colleagues evaluated the antihypertensive effect of oral
magnesium supplementation (22 mmol/d as magnesium aspartate) in
91 middle-aged and elderly women with mild to moderate
hypertension in a 6-month, double-blind, placebo-controlled
trial. Magnesium lowered systolic and diastolic BP by an average
of 2.7/3.4 mm Hg. When combining potassium supplementation and
salt restriction to magnesium supplementation, these workers
found a greater fall in BP (7.6/3.3 mm Hg). Brazilian researchers
reported that a 6-week course of magnesium oxide reduced BP in 15
hypertensive subjects by 5.9 mm Hg.
In other studies, oral magnesium supplementation has also
further lowered BP in the following subjects already receiving
antihypertensive drugs: a) hypertensive adults on beta-blocker
therapy given magnesium aspartate for up to 12 weeks; and b)
subjects taking diuretics, in whom magnesium lowered pressure and
increased tissue magnesium and potassium levels, and/or
Na+, K+ or ATPase activity.
Although these reports are suggestive, other clinical trials
have demonstrated no BP effects of magnesium supplementation. As
emphasized in a recent editorial, these controversies may not be
resolved no matter how many progressively larger or more
expensive clinical trials are performed, as long as hypertensive
subjects are considered as a homogeneous group, which they are
not, either pathophysiologically in general, or with respect to
their magnesium metabolism in particular.
Proper patient selection
Specifically, as early as 1983 we observed that despite a
uniform intracellular magnesium deficit, hypertensive subjects
exhibit a range of extracellular magnesium values, within the
normal range, that seem to be closely related to the activity of
pressor hormone systems such as the renin-angiotensin system.
Subjects with normal to high plasma renin activity (PRA) exhibit
lower serum magnesium values that predict a hypotensive response
to magnesium supplementation. Other hypertensive subjects who
have low PRA and/or salt sensitivity may exhibit no response or
even a mild pressor response to magnesium. Interestingly, such
patients may respond better to calcium supplements. Grouping all
subjects together according to only blood pressure levels as if
they were the same obscures the dramatic magnesium responses in
those with high PRA; the net result for the entire group might
appear modest or nil.
Hence, progress in this area will require a priori
identification of subgroups of patients on the basis of PRA,
dietary salt sensitivity, reproducible and sensitive measures of
extracellular magnesium, or other criteria useful in identifying
subjects for whom oral magnesium supplementation will be
particularly effective.
For now, all subjects should at least increase their magnesium
intake to achieve current RDA standards. Similar to current
recommendations for dietary salt restriction in hypertension,
magnesium supplementation beyond these levels should only be used
on a trial basis for 1 to 6 months and continued only if a
clear-cut response is observed.
Suggested reading
Itoh K, Kawasaki 1, Nakamura M. The effects of high oral
magnesium supplementation on blood pressure, serum lipids and
related variables in apparently healthy Japanese subjects. Br J
Nutr. 1997;78:737-750.
Kawano Y, Matsuoka H, Takishita S, Omae T. Effects of
magnesium supplementation in hypertensive patients:assessment by
office, home, and ambulatory blood pressures. Hypertension.
1998;32:260-265.
Paolisso G, Barbagallo M. Hypertension, diabetes mellitus, and
insulin resistance; the role of intracellular magnesium. Am J
Hypertens. 1997;1O:346-355.
Resnick LM. Magnesium in the pathophysiology and treatment of
hypertension and diabetes mellitus: where are we in 1997?
[editorial]. Am J Hypertens. 1997;10:368-370.
Q & A: Prescribing Oral Magnesium
Q: When do you suspect a magnesium
deficit?
DR. RESNICK:
A cellular magnesium deficit should be
suspected—independently of age—in subjects with
hypertension, coronary and peripheral atherosclerotic disease,
various cardiac arrhythmias, obesity, and or type 2 diabetes
mellitus.
Q: Do you recommend that patients take
supplemental magnesium?
DR. RESNICK: For the appropriate patient,
yes. The efficacy of supplementation depends on targeting
individuals who will benefit most from magnesium supplementation,
and providing an adequate, absorbable dose. Furthermore,
magnesium supplementation may be easier than increasing dietary
magnesium, which may involve lifestyle changes most patients find
difficult.
Q: Which hypertensive patients will most
likely benefit from magnesium supplementation?
DR. RESNICK: Magnesium supplementation is
most effective for lowering blood pressure (BP) in subjects whose
renin system is activated. These are the patients who respond
best to beta blockers, converting enzyme inhibitors, or
angiotensin-II blockers—drugs that block the secretion,
activation, or action of the renin angiotensin system.
Interestingly, these drugs may also increase serum magnesium
levels.
Q: What are the objectives of treatment
with oral magnesium?
DR. RESNICK: We include magnesium
supplementation early, both in the nondrug stage of therapy for
borderline or mild hypertension, and also together with drugs
before increasing medication dosage or adding another drug to the
regimen.
Q: Should magnesium be considered
routinely for patients on long-term diuretic therapy?
DR. RESNICK: Absolutely. Patients taking
diuretics, especially those that do not spare potassium and
magnesium, are at significant risk for magnesium depletion, which
may limit the efficacy of diuretic therapy. When magnesium
supplementation is added to antihypertensive regimens containing
diuretics, BP lowering may be enhanced, symptoms may improve, and
patients may feel better.
Q: What are common dosing
strategies?
DR. RESNICK: We suggest adding at least the
minimum recommended dietary allowance for magnesium
(approximately 200 to 400 mg/day elemental magnesium),
independent of dietary intake. Most patients take magnesium
tablets after meals. Diabetic patients, who are uniformly
magnesium depleted, receive the highest tolerated dosage.
Q: Is it worrisome that the latest
guidelines recommend raising dietary calcium and vitamin D
without commensurately increasing magnesium intake?
DR. RESNICK: For general health, hypertension
not withstanding, it’s important to increase dietary
magnesium commensurately with any increases in calcium and
vitamin D. For example, current data suggest that salt may
influence blood pressure only in subjects with concomitantly
lower calcium intakes, but not in those with adequate calcium
intakes. Similarly, a balance between calcium and magnesium
intake should be maintained. Increasing vitamin D beyond
approximately 400 UI/d also may be counterproductive. Higher
amounts, especially in combination with high calcium intakes, can
contribute to extraskeletal accumulation of calcium in the
cardiovascular system, urinary tract, and other soft tissue
sites, with potentially harmful results.
Q: Why do some clinical trials of
magnesium supplementation in hypertension show no
benefit?
DR. RESNICK: Hypertension is a heterogenous
phenomenon, not a uniform epidemiologically-defined condition. As
such, it needs to be treated specifically and individually, based
on the type of hypertension present. If all patients are lumped
together without attention to factors underlying the
hypertension, which may differ drastically from patient to
patient, the benefit of any single therapy in some patients may
be minimized or even obscured by the results in others in whom
this therapy is less relevant.
The above article is from the "The Magnesium Report",
First Quarter 1999. Blaine Pharmaceuticals is the
manufacturer of Mag-Ox 400 and Uro-Mag magnesium
supplements.

Go
to Blaine Pharmaceuticals
|
This page was first uploaded to The Magnesium Web Site on
October 14, 2002
http://www.mgwater.com/




